 |
Return to MOSDOH Modules Index
Module 2
Learning Objectives:
Definitions
Supragingival biofilm- a film of bacteria surrounded by extracellular polymeric substances (EPS) firmly attached to the teeth above the gum line (dental biofilm).
Subgingival biofilm- a film of bacteria surrounded by extracellular polymeric substances (EPS) firmly attached to the teeth below the gum line (dental biofilm). The biofilm is attached to the tooth surface in the sulcus and when the gingiva detaches from the tooth below the sulcus.
Plaque- the visible accumulation of a dental biofilm.
Calculus or tartar- mineralized plaque that can be supra- or subgingival.
Dental caries- demineralization of tooth enamel that reveals the dentin and can extend to the pulp resulting in pulpitis.
Periodontal disease- all disorders of the supporting structures of the teeth (periodontium). The periodontium includes the gingiva, periodontal ligament, and supporting alveolar bone. Periodontal disease includes gingivitis and periodontitis.
Gingivitis- is an inflammation of the gingiva only. The gingiva become edematous (swollen), erythematous (red) and will bleed.
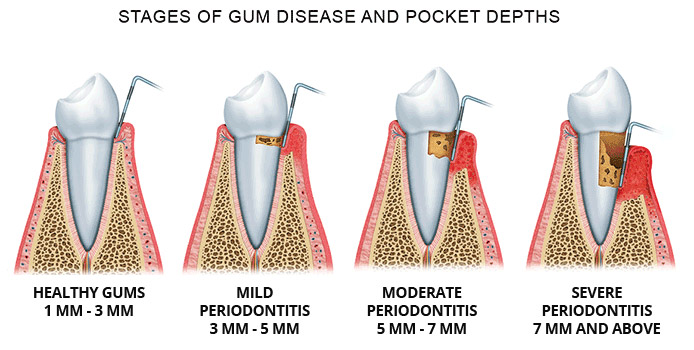
Periodontitis- is an infection and inflammation of the gingiva, periodontal ligament and alveolar bone. As the disease progresses the gingiva detach from the tooth surface forming periodontal pockets (see image below). In time the periodontal ligament is destroyed causing the tooth to be loose. If the inflammation becomes more severe destruction of the alveolar bone can also occur. In severe periodontitis the gingiva pulls away from the teeth, periodontal pockets form, the teeth loosen and fall out and alveolar bone loss occurs.
According to the Centers for Disease Control nearly half (47.2%) of adults aged 30 years and older have some form of periodontal disease. The prevalence of periodontal disease increases with age and 70.1% of adults 65 years and older have periodontal disease. Periodontal disease is the leading cause of tooth loss in adults. It is more common in men than women (56.4% vs 38.4%), adults living below the federal poverty level (65.4%), adults who have not graduated from high school (66.9%), and adults who smoke (64.2%).
Periodontal disease does not occur if supragingival and subgingival biofilm/plaque is routinely removed. No plaque no periodontal disease. Biofilm/plaque causes inflammation of the gingiva (gingivitis). The bacteria in the biofilm/plaque produce factors that breakdown the soft tissue of the gingiva and utilize it for nutrients. The breakdown of the gingiva causes the immune system of the host to respond. In attempting to eliminate the bacteria in the plaque more damage is done causing the gingiva to detach from the tooth. The plaque then grows down further below the sulcus (periodontal pockets) and causes more inflammation. The immune response and bacterial factors from the subgingival plaque cause destruction of the periodontal ligament. As the periodontal pockets go deeper alveolar bone is destroyed and lost.
Risk factors
Certain factors increase the risk for periodontal disease:
Timing of Plaque and Calculus Formation
A diverse collection of oral microorganisms are found in dental biofilms. A small sample of dental biofilm yields cultures containing between 12 and 27 bacterial species. After a thorough cleaning to remove dental biofilm on the tooth surface it only takes about 24 hours for dental biofilm to form again. If not removed by brushing two times a day and daily flossing the biofilm can accumulate to form plaque. If the plaque is mineralized it becomes calculus (tartar). Plaque formation and its mineralization to calculus takes on average about 12 days (range of 10 to 20 days). Calculus is difficult to remove and REQUIRES professional cleaning to remove it. If plaque and calculus are not regularly removed patients are much more likely to develop periodontal disease.
Periodontal disease
Gingivitis
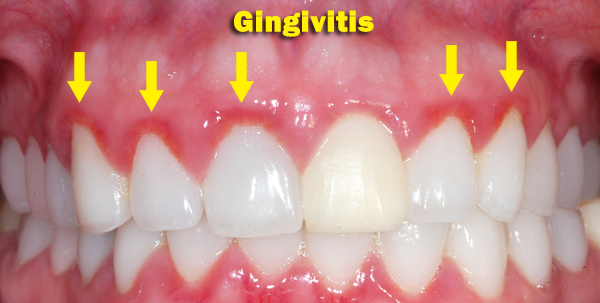 |
Patient with Gingivitis http://diseasespictures.com/wp-content/uploads/2016/09/gingivitis-4.png |
The more subgingival biofilm/plaque that accumulates on the teeth the more likely a patient will develop gingivitis. Gingivitis does NOT occur following any specific pathogen being present in the dental biofilm/plaque. More anaerobic Gram negative rod-shaped bacteria and spirochetes are present in the subgingival biofilm/plaque when compared to supragingival biofilm/plaque. When the numbers of anaerobic Gram negative rod-shaped bacteria and spirochetes increase compared to the other bacteria in the subgingival biofilm/plaque then gingivitis is more likely to occur.
The subgingival biofilm/plaque bacteria produce proteases and hemolysins to obtain nutrients by damaging host tissues. As a result, the host immune response produces an inflammatory reaction in the gingiva. Inflammation is manifest in the gingiva by swelling (edema), redness (erythema) and bleeding following brushing/flossing. Rarely, if ever, are the gingiva painful. If they are the patient may state that their gums are tender. If the biofilm/plaque is not removed the gingiva may detach from the tooth and allow the biofilm/plaque to extend further down the tooth’s surface.
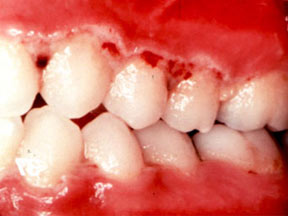
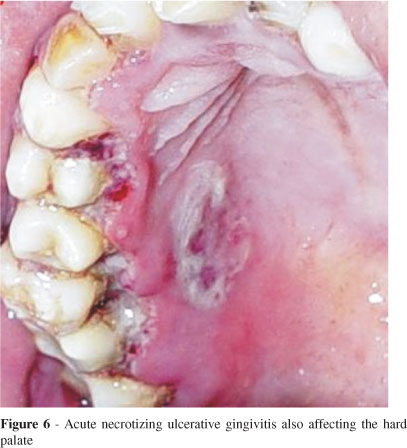
Necrotizing Ulcerative Gingivitis
 |
 |
Patient with NUG http://www.emeryvilledental.com/library/7379/ANUGTrenchMouth.html |
Patient with NUG with an ulcer http://www.scielo.br/scielo.php?script=sci_arttext&pid=S1807-59322007000600018 |
A severe form of gingivitis called necrotizing ulcerative gingivitis or trench mouth has a rapid onset. It is relatively uncommon and is more likely to occur in patients with poor oral hygiene, those that are immunosuppressed, those who smoke or patients under physical and/or emotional stress (e.g,. boot camp, college, soldiers in combat).
Symptoms include fever, malaise, lymphadenopathy, halitosis, and painful and inflamed gingiva with necrotic gray to black ulcerations (crateriform lesions) of the interdental papillae that bleed easily (papillae are blunted and no longer cone shaped). A gray pseudomembrane covers lesions and is easy to remove. The dental biofilm/plague in these patients usually contains a lot of Fusobacterium spp., Prevotella intermedia, and especially spirochetes (Treponema denticola).
If not treated trench mouth can spread to the oropharynx causing Vincent’s angina. Vincent’s angina can occur rapidly (hours to days). Symptoms include; fever, swelling of lower face and neck, difficulty with speech or swallowing, membranous pharyngitis, painful ulcers on gingivae, buccal mucosa, and pharynx with hyperemic patches, and edema.
Periodontitis
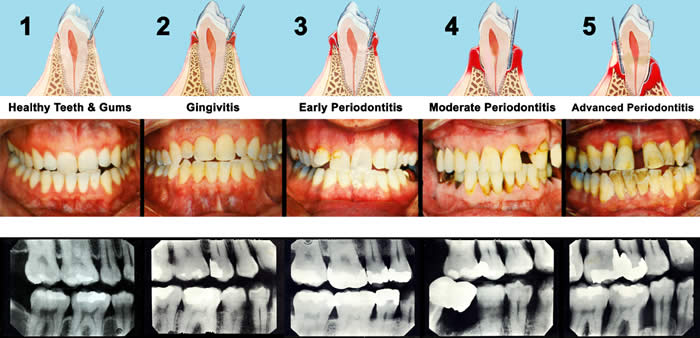 |
http://mizar5.com/Periodontal_Images.png
Subgingival biofilm/plaque is also an important cause of periodontitis. Unlike gingivitis, certain bacteria if present in the subgingival biofilm/plaque make periodontitis more likely. The risk of periodontitis developing depends on the bacteria present in the subgingival biofilm/plaque. These bacteria have been placed in complexes based on their likelihood of causing periodontitis.
Chronic periodontitis
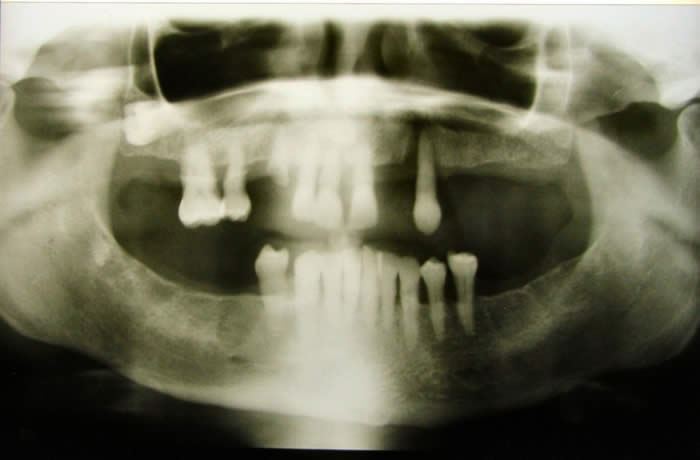 |
Radiograph of Patient with Severe Periodontitis and Tooth Loss |
Periodontal disease is usually a chronic disease taking many months to develop.
Red complex bacteria (e.g., Porphyromonas gingivalis, Tannerella forsythia, and Treponema denticola) if present in large numbers in the subgingival biofilm/plaque place a patient at high risk of developing localized periodontitis.
Orange complex bacteria (e.g., Fusobacterium nucleatum, Prevotella intermedia, Prevotella nigrescens, Parviomonas micra, Eubacterium nodatum, and various Camplylobacter species (C. rectus) if present in large numbers in the subgingival biofilm/plaque place a patient at moderate risk of developing localized periodontitis.
Yellow complex bacteria (e.g., Streptococcus intermedius, Streptococcus sanquinis, Streptococcus oralis, Streptococcus mitis, and Streptococcus gordonii) if present in large numbers in the subgingival biofilm/plaque place a patient at low risk of developing localized periodontitis.
Aggressive Periodontitis
Aggressive periodontitis is an uncommon rapid detachment of the periodontal ligament and destruction of alveolar bone. It usually affects younger patients < 30 years of age (particularly adolescents) that appear otherwise healthy. It is associated with phagocyte abnormalities and hyper-responsive macrophages leading to inflammation of the tissue. The plaque microflora in aggressive periodontitis is similar to chronic periodontitis. However, the presence of certain bacteria in the biofilm/plaque increases the risk for this disease to develop (Aggregatibacter actinomycetemcomitans or Eikenella corrodens in concert with Fusobacterium nucleatum). Sparse amounts of biofilm/plaque are generally found relative to degree of periodontal destruction.
There are two forms of aggressive periodontitis based on clinical presentation.
Prevention and Treatment
Gingivitis can be prevented and treated with good oral hygiene and routine professional cleaning.
Periodontal disease can be prevented with good oral hygiene and routine professional cleaning.
Periodontal pocketing- Water Irrigation- when deep periodontal pockets form and do not resolve the use of a water irrigator can lower the inflammation in the gingiva. It appears to do this by cleansing food particles and dead tissue from the periodontal pockets and by oxygenating the subgingival plaque and the tissues surrounding the pockets. Oxygen can kill or slow the growth of anaerobic bacteria present in the subgingival plaque.
Severe periodontal disease treatment may include scaling and root planing followed by antiseptic chip*, antibiotic gel** or antibiotic microspheres*** placement, oral antibiotics (metronidazole and oxacillin) and surgery (flap surgery, tissue grafts, bone grafts).
NUG treatment- Metronidazole (targets the obligate anaerobes) with a non-steroidal anti-inflammatory and anti-pain medication (e.g., ibuprofen).
Aggressive periodontitis- Metronidazole (targets the obligate anaerobes) plus amoxicillin (to kill facultative anaerobe A. actinomycetemcomitans).
* Antiseptic chip- gelatin containing chlorhexidine placed in pockets after scaling and root planing.
** Antibiotic gel- a gel containing doxycycline placed in pockets after scaling and root planing.
*** Antibiotic microspheres- small particles containing minocycline placed in pockets after scaling and root planing.
Test yourself to see what you have learned.
Return to MOSDOH Modules Index
References:
Eke PI., Dye BA., Wei L., Thornton-Evans GO., and Genco RJ. 2012. Prevalence of Periodontitis in Adults in the United States: 2009 and 2010. J Dent Res. 91: 907-908 doi:10.1177/0022034512457373
Hasan A., Palmer RM., 2014. A clinical guide to periodontology: Pathology of periodontal disease. Brit Dent J. 216:457-461. doi:10.1038/sj.bdj.2014.299
Al-Ghutaimel H., Riba H., Al-Kahtani S., and Al-Duhaimi S., 2014. Common Periodontal Diseases of Children and Adolescents. Int J Dent, Article ID 850674, 7 pages. doi:10.1155/2014/850674
© 2016 Neal R. Chamberlain,Ph.D., and Priscilla L. Phillips, Ph.D. All rights reserved.
Site Last Revised 10/13/16
Neal Chamberlain, PhD. and Priscilla L. Phillips, Ph.D. A. T. Still University of Health Sciences/Kirksville College of Osteopathic Medicine.
Take Care and Think Microbiologically!
Disclaimer:
None of the contributors to this web site assume liability for any use or misuse of the information provided here. This information does not replace dental/medical consultation, but is intended for EDUCATIONAL purposes only. Any mention of commercial products is for inclusive purposes and is not an endorsement. Every effort has been made to provide accurate information about the products mentioned in this web site, but consult product labels for the most up-to-date information available. This web site does not constitute a medical diagnosis; seek advice from a dentist/physician (if you are not one yourself) before making a diagnosis and/or undertaking any of the treatments suggested here.