|
|
|
|
Carditis, or inflammation of the heart, is most conveniently broken
down into three categories:
Pericarditis - Inflammation
of the pericardium
Myocarditis - Inflammation
of the heart muscle
Endocarditis - Inflammation of
the endocardium
PERICARDITIS
Pericarditis, inflammation of the fibroserous sac enclosing the heart, manifests itself as one of three types as a result of the bodies reaction to the infecting agent:
Acute serofibrinous pericarditis - the result of
virus infection
Acute purulent pericarditis - the result of bacterial
infection (except for infection by
Mycobacterium tuberculosis)
Chronic pericarditis - the result of infection by
M.
tuberculosis or fungi
ETIOLOGY:
Virtually any infectious agent that reaches the pericardium
is capable of causing pericarditis. The most common viruses causing the
disease are members of the Picornaviridae (enteroviruses) while the most
common bacteria infecting the pericardium are the pyogenic cocci (Staphylococcus,
Streptococcus,
Neisseria).
Chronic pericarditis is most commonly caused by M. tuberculosis
or Histoplasma capsulatum.
OVERVIEW OF DISEASE:
In most cases the infecting microorganism reaches
the pericardium via the circulatory system. Rarely the organism will enter
the pericardium by direct extension from the lung or direct inoculation
during surgery, invasive medical procedures or trauma. The organism colonizes
the pericardium and stimulates an inflammatory reaction which can result
in destruction of heart tissue. If the infecting organism is a virus there
is also viral cell lysis. The inflammatory reaction results in the accumulation
of serous or purulent exudate which may in turn cause cardiac tamponade
(the exertion of pressure or compression on the heart) and circulatory
failure.
PATHOLOGY:
Microorganisms reach the pericardium by the blood, lymph, direct extension from pulmonary or myocardial foci of infection, or direct inoculation during surgery, other invasive procedures, or penetrating trauma. Viral infections are usually blood-borne and often infect the myocardium as well as the pericardium. Pneumococci or other primary pulmonary pathogens usually infect the pericardium by extension from an adjacent pneumonitis; the common causes of bacteremias - staphylococci, meningococci and H. influenzae - are more likely to reach the pericardium through the blood-stream. A pre-existing, noninfectious pericarditis, as is seen with uremia or following cardiac surgery, may increase susceptibility to hematogenous bacterial pericarditis.
Acute serofibrinous pericarditis. The viruses typically causing this disease produce a relatively mild inflammatory reaction that is associated with focal damage to the adjacent myocardium. The response varies from a small amount of serous fluid with mononuclear cells and fibrinogen to a large, neutrophil-rich, bloody effusion. The tissue damage is the result of:
1.
Direct cellular damage by the infecting virus
2.
Destruction of viral-infected cells by sensitized T-lymphocytes
3.
Antibody-dependent, cell-mediated cytotoxicity (null cell-dependent).
Mild fibrosis and occasional adhesions between visceral and parietal surfaces may mark the healing of viral pericarditis. However, such a fibrotic reaction rarely gives rise to a constrictive pericarditis. The disease is self limiting and rarely fatal.
Acute purulent pericarditis. The bacteria typically causing this disease produce a relatively strong rapidly progressing purulent reaction. The purulent material contains large numbers of polymorphonuclear leukocytes in a large volume of effusion. The tissue damage is the result of:
1.
Toxin and enzyme production by the bacteria
2.
Myocardial damage
3.
Rapidly progressing cardiac tamponade
Healing is associated with extensive fibrosis that may progress to a chronic, constrictive pericarditis. The mortality rate exceeds 50%.
Chronic pericarditis. This is most commonly
caused by M. tuberculosis. About 5% of patients with pulmonary tuberculosis
will have pericardial involvement. The early granulomatous stages are associated
with large pericardial effusions (> 300 ml) that are typically serosanguinous
and contain a predominance of mononuclear cells. As the disease evolves,
the inflammatory process becomes chronic; fusion of the parietal and visceral
pericardium may result, yielding constrictive pericarditis and circulatory
failure.
CLINICAL SYMPTOMS AND SIGNS:
Acute serofibrinous pericarditis gives rise to:
a.
Chest pain - rapid in onset, persistent for several hours to days. It is
worse during
inspiration and recumbency but improves with leaning forward. The pain
is usually in the
upper abdominal region overlying the stomach and may be sharp, dull, constricting
and/or
crushing making clinical differentiation from myocardial infarction difficult.
Radiation
of
pain to the left trapezius ridge is particularly characteristic.
b. Fever - not prominent
c. Malaise
d.
Pericardial friction rub - this is usually a triphasic sound that resembles
scratching or the
crunch of footsteps on cold snow.
Acute purulent pericarditis gives rise to:
a. Little, if any, chest pain
b. Prominent fever and chills from the severe underlying infection
c. Cardiac tamponade which leads to:
(1) Dyspnea
(2) Agitation
(3) Orthopnea
(4) Cough
d. Pericardial friction rub - occurs in less than 50% of patients.
Chronic pericarditis is generally due to M. tuberculosis and the symptoms are those of tuberculosis. These include:
a. Fever
b. Night sweats
c. Weight loss
d. Fatigue
e. Progressive circulatory failure due to:
(1) Slowly progressing dyspnea
(2) Ascites
(3) Edema
In general all types of pericarditis may result in:
a.
Abnormal heart sounds other than those due to pericardial friction
b.
Pericardial effusion that causes:
(1) Decreased or muffled heart sounds
(2) An area of dullness to percussion at the tip of the left scapula (Ewart's
sign)
(3) Pulsus paradoxes
(4) Cervical venous pressure increase with inspiration (Kussmaul's sign)
(5) Abnormal EKG
TREATMENT:
The treatment includes bed rest, control of pain
with non-steroidal anti-inflammatory agents and anti-microbial therapy
based on the species of the infecting agent and its sensitivity pattern.
MYOCARDITIS
ETIOLOGICAL AGENTS:
Many species of viruses, bacteria, chlamydia, rickettsia,
fungi and protozoans can cause myocarditis. However, viruses are the most
important infectious agents. Of these, the enteroviruses are the single
most important group.
OVERVIEW OF DISEASE:
The disease is an infection of the myocardium or
muscle of the heart. The virus is ingested in fecally-contaminated water
and/or food and eventually, either directly or indirectly, reaches the
heart. There may be a prior skin infection before heart effects are seen.
The virus invades the heart muscle cells and causes necrosis of the cells
and clinical effects.
PATHOLOGY:
Necrosis of myofibers is seen initially and this may be either patchy or diffuse. An acute inflammatory reaction with polymorphonuclear leukocytes may be seen during the first few days of infection, and this is promptly followed by a mononuclear leukocyte cellular infiltrate by the end of the first week. Late changes include fibrosis and loss of myofibers.
Myocardial cell damage appears to occur in several ways:
a.
Direct viral damage (e.g., with the Coxsackie B virus).
b.
Inhibition of humoral and macrophage-mediated immunity early in the infection.
c.
T-cell mediated cell lysis late in the disease.
DIAGNOSIS:
In infants and young children the disease progresses rapidly, beginning with fever, tachycardia and listlessness and progressing with signs and symptoms of CNS, gastrointestinal, liver and myocardial involvement. Cardiac failure may be evident within a few days of onset of the illness.
In older children and adults the illness usually progresses more slowly with generalized manifestations of viral infections localized to:
a. Respiratory tract
(1) Moderate fever
(2) Cough
(3) Coryza
b. Abdomen
(1) Pain
(2) Nausea
(3) Vomiting
Myocardial involvement becomes manifest one or two weeks after the initial illness with:
a.
Fever
b.
Malaise
c.
Fatigue
d.
Dyspnea (labored or difficult breathing)
e.
Palpitations (tachycardia)
f.
Altered heart sounds (muffled, transient pericardial rubs)
g.
Chest pain
h.
Electrocardiographic changes
i.
Cardiomegaly
j.
Pulmonary congestion
k.
Abnormal laboratory findings:
(1) Increased sedimentation rate
(2) Increased leukocyte count
(3) Increased myocardial isoenzymes
(a) Serum Glutamic Oxaloacetic Transaminase (SGOT)
(b) Creatinine Phosphokinase (CPK)
(c) Lactic Dehydrogenase (LDH)
TREATMENT:
Viral myocarditis is typically a mild disease and
responds well to bed rest. Bacterial, fungal and protozoan myocarditis
can be treated with the appropriate antibiotics. Glucosteroids and other
immunosuppressive drugs are CONTRAINDICATED.
ENDOCARDITIS
Endocarditis, inflammation of the membrane lining
the chambers of the heart and covering the cusps of the various valves,
is caused directly by microbial colonization of the endocardium or indirectly
by induction of autoimmunity, as in acute rheumatic fever. Direct colonization
is termed infective endocarditis and is caused by microorganisms physically
present in endocardial lesions know as vegetations. The disease
may be either acute or chronic.
ETIOLOGY:
Almost all bacteria and many fungi, when they get
into the blood stream, can cause infective endocarditis. However, 80% of
the cases are caused by streptococci or staphylococci.
OVERVIEW:
Relatively avirulent microorganisms derived from
the normal flora of the body cause most cases of infective carditis. They
gain access to the blood intermittently, as a result of minor trauma to
the mucosa of the oropharynx, gastrointestinal tract or genitourinary tract.
Such transient bacteremias usually occur without ill effects but they will
lead to endocarditis in patients with an underlying cardiovascular lesion
or with a suppressed immune system. Intravenous drug abusers commonly have
infective endocarditis due to Staphylococcus aureus from contaminated
needles. Blood-borne organisms are deposited on the downstream side of
the valves where they colonize and cause disease.
PATHOLOGY:
As bacteria colonize the endocardium, they form vegetations which vary in size from tiny bodies to masses large enough to occlude valve orifices. Often, they are soft and friable and only loosely attached to the endocardium. Thus, they break off easily to form arterial emboli. Fungal vegetations tend to be bulky, giving rise to large emboli. Apart from the propensity to generate emboli, there is no correlation between size of vegetation and severity of endocarditis.
The bulk of the vegetation is an amorphous mass of fibrin and platelets containing colonies of microorganisms. There may be inflammatory cells attached to the vegetation. There are four consequences to the formation of this vegetation:
a. Organisms within the vegetation are protected from antibodies, complement, and leukocytes
b. Organisms within the vegetation
are metabolically inactive, replicating at an unusually slow
rate, rendering them relatively resistant to the action of many antimicrobics
c. Healing is slow because
macrophages and fibroblasts must spread through the vegetation
and endothelial cells must grow over the surface.
d. Emboli are generated when vegetations break off; these cause infarcts.
Abscesses may develop by direct invasion of the valve
rings of the heart near the vegetations. These are common with pyogenic
cocci but rare with other organisms.
CLINICAL SYMPTOMS:
The interval between the colonization of the endocardium and the onset of symptoms is two weeks. Death can occur about 6 weeks after colonization if the disease goes untreated. The initial symptoms are those of any infection:
a. Low grade fever
b. Anorexia
c. Fatigue
d. Weight loss
e. Anemia
f. Splenomegaly
Serologic findings include:
a. Hypergammaglobulinemia
b. High levels of rheumatoid
factor
c. High levels of antinuclear
antibody
d. High levels of circulating
immune complexes
The circulating immune complexes give rise to skin manifestations which include:
a. Osler's nodes
b. Splinter hemorrhages
c. Petechia
d. Janeway's lesions
e. Roth spots
Urinary findings may include:
a. Proteinuria
b. Microscopic hematuria
c. Red blood cell casts
Heart murmur is present at some times during the
disease but is generally not constant. If the tricuspid valve is damaged
to the extent that there is tricuspid regurgitation, Carvallo's sign will
be present; this is augmentation of the pan-cystolic murmur by inspiration.
DIAGNOSIS:
Because the clinical features of the disease can
be quite variable and often nonspecific, diagnosis is mainly based on laboratory
tests. Blood culture and serologic testing are the most important. Always
use venous blood to isolate the organism. A positive blood culture with
some or all of the symptoms listed is needed to obtain the diagnosis.
TREATMENT:
Antibiotic therapy must persist for at least 14 days, even if symptoms disappear prior to that time. A combination of antibiotics, rather than a single antibiotic, is always used. If no organism has been isolated after repeated attempts the recommended therapy is:
Ampicillin, given IV every 4 hours + Gentamycin, given every 8 hours.
If an organism has been isolated, then the antibiotic regimen is based on the species of the etiologic agent, the age of the patient and the extent of the disease. The regimens are complex and are listed in various reference books.
If antibiotic therapy is not successful surgical removal of infected endocardium may be necessary. This is especially true with fungal infections and when the patient has an intracardiovascular prosthesis. Nonspecific therapy includes:
Bed rest
Low salt diet
Alleviation of congestive heart failure
Control of fever
Transfusion to maintain a hematocrit close to 30%
Anticoagulation with heparin is CONTRAINDICATED.
MYOCARDIAL INFARCTION
ETIOLOGY:
Chylamydia pneumoniae, a Gram-,
pleomorphic, obligate intracellular parasite.
OVERVIEW:
Myocardial infarction can occur in the absence of
the common risk factors such as hypercholesterolemia, diabetes mellitus
or cigarette smoking. The sequence of events that leads to acute myocardial
infarction includes atherosclerotic plaque formation, plaque rupture, coronary
artery thrombosis and coronary occlusion. Anything that leads to plaque
rupture can result in myocardial infarction. C. pneumoniae infection
is one of those risk factors that can induce plaque rupture. The plaque
debris lodges in a blood vessel upon rupture and blocks blood flow.
PATHOLOGY:
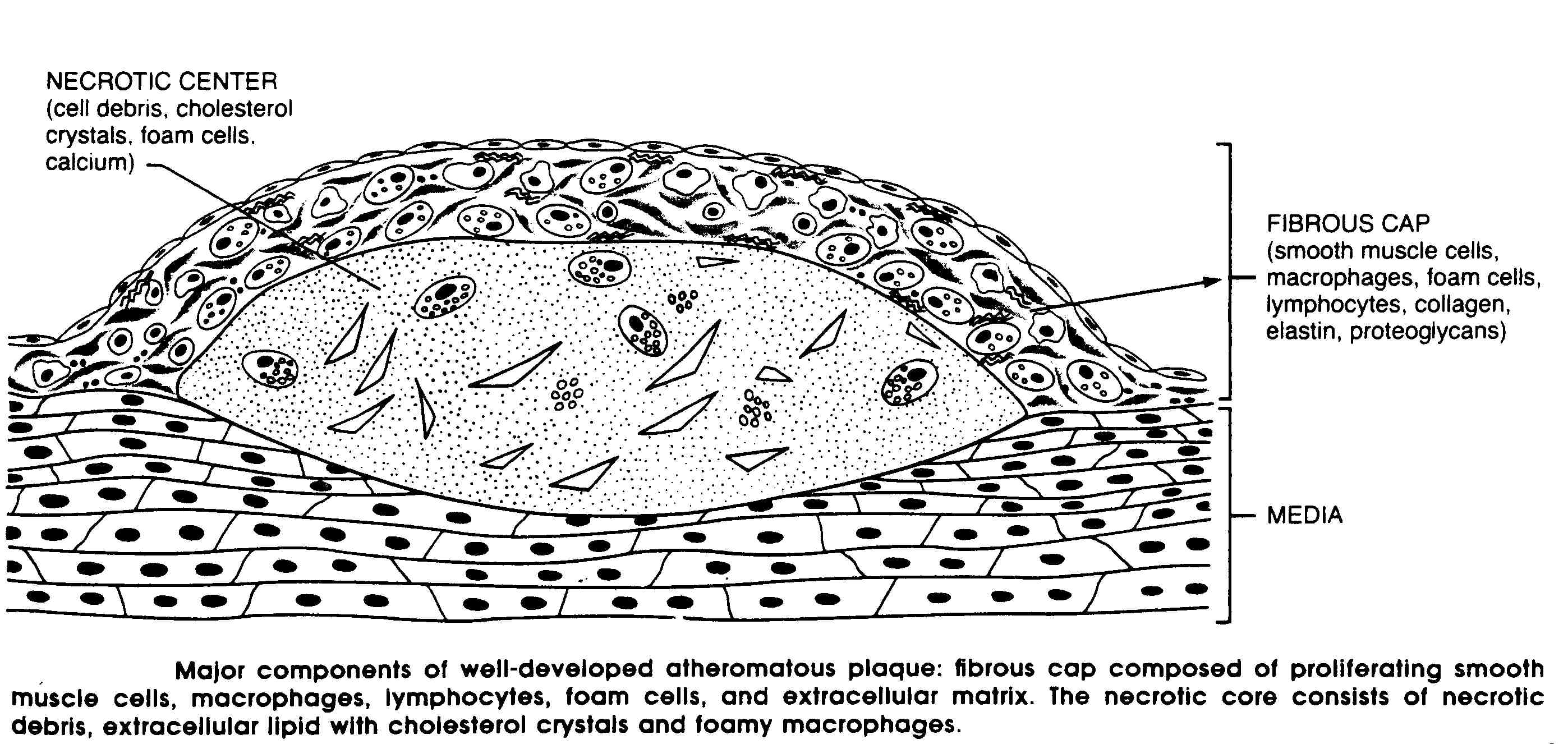
The key pathologic features in atherosclerosis are focal intima thickenings and lipid accumulation, producing the characteristic atheromatous plaques. This is a white to yellowish/white body impinging on the lumen of the artery. On section, the superficial portion of these lesions at the luminal surface tends to be firm and white (the fibrous cap) and the deep portions yellow or whitish yellow and soft. The centers of larger plaques may contain a yellow, grumous debris called an atheroma.

C. pneumoniae is present in the fibrous cap
if myocardial infarction is due to an infectious process.
SYMPTOMOLOGY:
The symptomology of myocardial infarction is the same in the non-infectious and infectious disease. The most common presenting complaint is pain which is described as
Severe (often the worst pain the patient has ever
experienced)
Deep and visceral
Heavy
Squeezing
Crushing
Typically the pain involves the central portion of
the chest and/or epigastrium and in about 30% of cases it radiates to the
arms. Fifteen to twenty percent of patients suffer no pain.
DIAGNOSIS:
The combination of substernal chest pain, persistent for more than 30 minutes and diaphoresis strongly suggests acute myocardial infarction. Laboratory findings include:
1. Non-specific indices of tissue necrosis and inflammation
a. Polymorphonuclear leukocytosis
b. Erythrocyte sedimentation rate that rises more slowly than the WBC count
2. The electrocardiogram
3. Serum enzyme changes
a. Creatine phosphokinase (CK)
b. Lactic dehydrogeinase (LDH)
4. Cardiac imaging
5. Presence of chlamidia in the plaque
6. Presence of antibiodies
to C. pneumoniae
TREATMENT:
Treatment for the infection is administration of one of these compounds (listed in order from most effective to least efficatious):
Macrolide antibiotics (erythromycin, azithromycin,
clarithromycin)
Tetracyclines
Fluoroquinolones (ciprofloxacin, norfloxacin and
ofloxacin)
|
|
|
|