MM 237-240, 619-621; ID 736-741, 747-752, 763-768, 721-723
Invasive Gastroenteritis
SHIGELLOSIS
OVERVIEW:
Shigellosis is primarily a disease of direct anal-oral transmission, although food-borne and water-borne and insect-borne epidemics have occurred. Despite modern environmental hygiene shigellosis persists because
1. Shigella are present in large numbers around the bases of toilets used by infected persons.
2. They readily pass through toilet paper onto the fingers.
3. As few as 2000 ingested organisms can cause disease.
4. It can be spread by horseflies
5. Closed population groups
often have substandard sanitation (e.g., prisoner-of-war camps,
homes for mentally retarded, Indian
reservations).
ETIOLOGICAL AGENTS:
Fifty Shigella species which fall into one of four serological groups.
Group A - S. dysenteriae (S. shiga) which is not encountered in the U.S. except for rare instances of imported disease.
Group B - S. flexneri, a species commonly isolated in the U.S.
Group C - S. boydii and related species which are rarely isolated in the U.S.
Group D - S. sonnei, the most commonly isolated cause of shigellosis in the U.S.
Shigella are gram-negative rods whose natural and sole habitat is the intestine of humans and other primates.
Shigellae are closely related to Escherichia coli and share antigens and toxin-producing capability with them. They produce both an endotoxin and an exotoxin. The exotoxin is an enterotoxin that produces diarrhea like the E. coli LT toxin and the cholera toxin. Acting as a neurotoxin, this exotoxin causes meningismus and coma. It can also cause an ulceration of the intestine. It results in mucous secretion. Upon invasion of a cell, shigellae produce and excrete another major virulence factor, NAD glycohydrolase, which destroys all of the NAD of the human cell and virtually shuts down cellular metabolism, causing cell death.
Shigellosis is mainly a disease of children between 1 and 4 years of age. It is transmitted by the 4-F's: food; fingers; feces; flies.
PATHOLOGY:
Upon ingestion the microorganisms pass to the large intestine where they must penetrate the cells of the epithelial lining in order to cause diarrhea. Non-invasive shigellae are not pathogenic. Non-toxin producing shigellae are also not pathogenic. Pathogenesis requires invasion and toxin production. Following intracellular penetration, multiplication occurs in the submucosa or lamina propria. Distortion of crypts occurs as clumps of cells are sloughed. This causes blockage. The accumulation of inflammatory cells behind the obstruction leads to the formation of microabscesses. Through spread and coalescence, larger abscesses form. Long sections of affected colon or sigmoid may be covered by a fibrous exudate containing huge numbers of neutrophils. Bleeding occurs from superficial ulcerations that are about 5 mm in diameter. Perforation is NOT a complication because of the superficial localization of the infection. Invasion of the blood stream is uncommon.
SYMPTOMS:
After an incubation period of 36-72 hours, the initial non-specific symptoms of fever and cramping abdominal pain are prominent. Diarrhea usually appears after 48 hours, with dysentery supervening about 2 days later. Abdominal tenderness is usually general, and the abdominal wall is not rigid. Sigmoidoscopy reveals intense hyperemia, multiple small bleeding sites, loss of transverse mucosal folds and thick, purulent mucous secretions. Tenesmus is present and the feces are bloody, mucoid and of small volume.
Fluid and electrolyte loss may be quite significant, particularly in pediatric and geriatric populations. There may be a peripheral neurites and convulsions. Escherichia coli septicemia may be initiated by shigellosis.
DIAGNOSIS:
Presumptive diagnosis is based on the acute onset of fever and diarrhea with bloody and mucoid feces. Definitive diagnosis requires the isolation of Shigella from the feces. Microabscesses in a rectal biopsy are suggestive of shigellosis. Diffuse involvement of the mucosa with multiple shallow ulcers 3-7 mm in diameter is suggestive of shigellosis. A rectal swab of an ulcer will reveal clumps of neutrophils, macrophages and erythrocytes.
Shigella are commonly isolated on S-S agar (Salmonella - Shigella agar).
TREATMENT:
Shigellosis is usually a
self-limited disease. However, fluoroquinolones (noraflacacin, ciprofloxacin,
ofloxacin) will kill S. dysenteriae and S. flexneri. S.
sonnei is best treated with aminoglycosides. Fluid and electrolyte
replacement is necessary in severe cases. Antidiarrheal compounds which
inhibit peristalsis are CONTRAINDICATED.
TYPHOID FEVER
OVERVIEW:
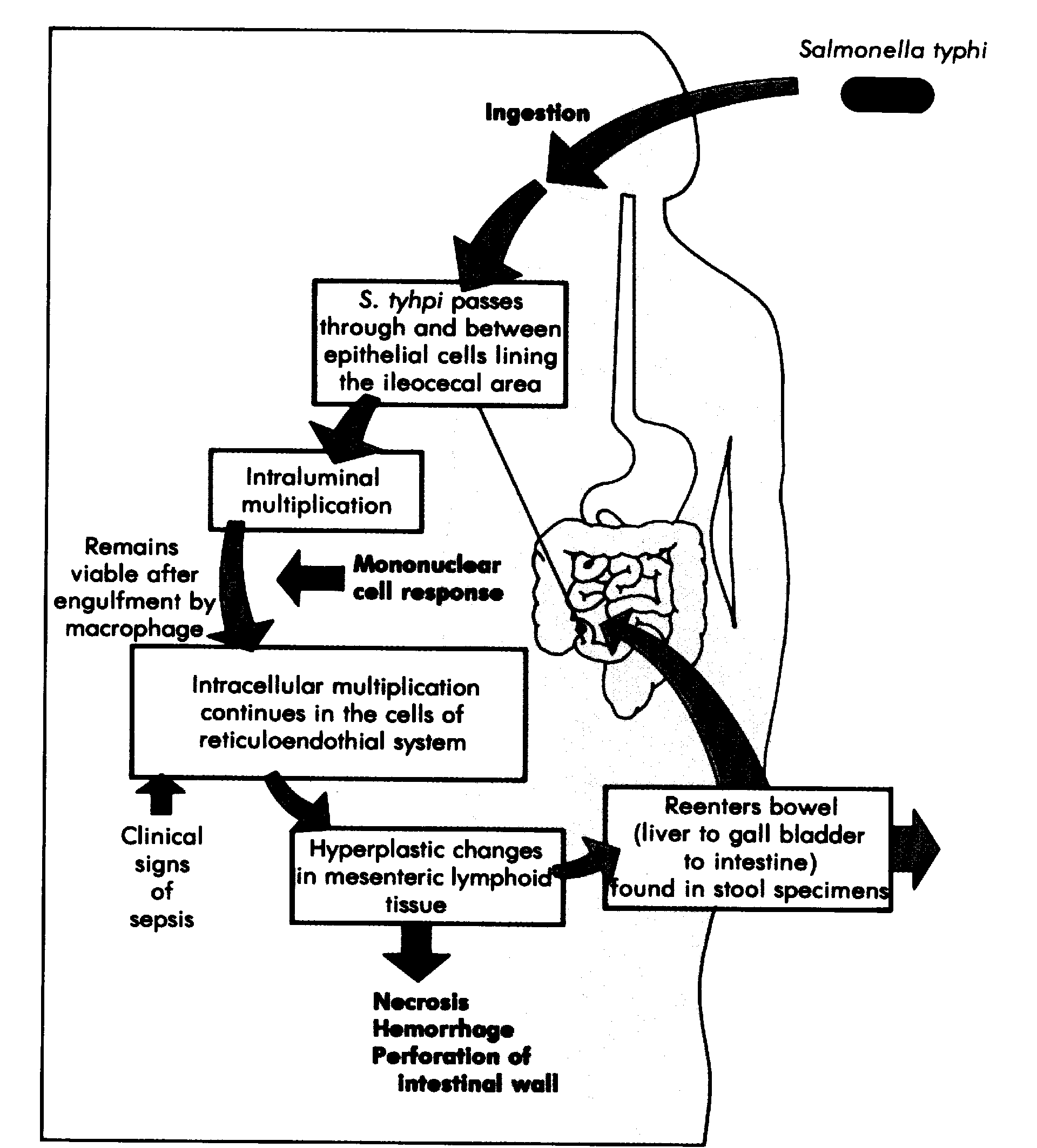
The sole source of S. typhi is the human intestinal tract. The organism is ingested in fecally-contaminated food or water. It passes to the duodenum where the organism rapidly penetrates the intestinal mucosa into the regional lymphatics, where it is phagocytized. Neutrophils are unable to kill ingested bacteria and they gain entry to the circulatory system. The resulting septicemia is quickly terminated through removal by reticuloendothelial cells in liver, spleen, bone marrow and lymph nodes. However, the bacteria multiply within these cells giving rise to a sustained bacteremia. During this sustained bacteremia; the biliary tract is infected and the organisms are excreted in the bile which seeds the intestinal tract with millions of bacteria, setting up a cycle of infection. In 14% of people infected, there is asymptomatic colonization of the gall bladder, with these people becoming carriers who spread the disease. The major virulence factor of S. typhi is the Vi antigen, a surface antigen which interferes with phagocytosis and, if the cell is still phagocytized, inhibits the respiratory burst which is instrumental in killing ingested bacteria. Endotoxin is another virulence factor.
ETIOLOGICAL AGENT:
The cause of typhoid fever
is Salmonella typhi. This is a cytophilic, gram-negative, aerobic,
noncapsulated, non-spore-forming, motile rod. Presumptive identification
is based on serological tests for the somatic (0) and flagellar (H) antigens.
PATHOLOGY:
The proliferation of large mononuclear cells derived from reticuloendothelial tissue is the most prominent feature of the pathology of typhoid fever. Involvement of lymphoid tissue in the intestinal tract, principally Peyer's patches in the terminal ileum, may lead to necrosis and ulceration at these sites. The erosion of blood vessels in the lesions may give rise to intestinal hemorrhage. Although intestinal lesions are usually confined to the mucosa and submucosa, muscular and serosal layers are occasionally penetrated, leading to intestinal perforation.
The liver is enlarged during typhoid fever; there are focal areas of necrosis with cloudy swelling of hepatic cells. The spleen and mesenteric lymph nodes are enlarged, and there is hyperplasia of reticuloendothelial cells. Bronchitis is common and pneumonia is not unusual. The maculopapular skin lesions are infiltrated with mononuclear cells, and there is vascular congestion. Overall, the tissues sharing major involvement are: kidney, spleen, bile duct, gall bladder, lymph nodes, lungs, liver, skin, bronchi and brain.
SYMPTOMS:
After an incubation period of 8-14 days, fever is usually the earliest indication of typhoid fever, rising in a step wise fashion during the first week. The onset of symptoms is gradual; they include:
anorexia
lethargy
malaise
general
aches and pains
dull,
continuous headache usually confined to the frontal regions
nonproductive
cough
nosebleed
(10% of patients)
vague
abdominal pain and discomfort
constipation
(20% of patients have mild diarrhea)
During the physical exam at this stage of the disease there is tenderness upon palpation of the lower quadrants of the abdomen. Distension is frequently present, and the examiner may experience a sensation of displacing loops of bowel filled with air and fluid on palpation of the abdomen.
During the second week of illness the temperature is sustained at 104° F. The patient is frequently severely ill during this phase of illness; weakness, mental dullness or even delirium may be prominent. Abdominal discomfort and distension increase, and diarrhea is more common than during the first week. The feces may contain blood.
The face is dull and expressionless. Rhonchi and scattered moist rales occur in as many as 50% of patients. Splenomegaly and rose-colored spots on the abdomen are common. These are small maculo-papular lesions that blanch on pressure. The lesions are usually sparse, not exceeding 20.
As the illness extends into the third week, the patient continues to be febrile and becomes increasingly exhausted and weak. If no complications occur, the patient may begin to improve toward the end of the third week. The temperature gradually begins to decline and may reach normal levels by the end of the fourth week.
DIAGNOSIS:
Typhoid fever may be confused with a wide variety of infections diseases characterized by fever. A history of travel in endemic areas or a prolonged febrile illness with or without typical manifestations of typhoid should arouse suspicion. Rose colored spots on the abdomen which persist for 2-4 days and recur in crops is the best diagnostic clue. Examination of blood reveals:
anemia
leukopenia with a relative decrease in polymorphonuclear leukocytes and
an absence of eosinophils
presence of S. typhi which can be isolated on S-S agar.
positive Widal reaction - this is agglutination of somatic and flagellar
antigens
Examination of feces reveals:
bloody feces
melena
presence of S. typhi
leukocytes
Examination of urine reveals:
proteinuria
presence of S. typhi
Confirmative diagnosis requires the isolation of S. typhi or the demonstration of a four-fold or greater rise in antibody to the "O" antigen.
TREATMENT:
The drug of choice is ciprofloxacin. Secondary antibiotics includes ceftriaxon. Laxatives and enemas are CONTRAINDICATED because of the danger of inciting intestinal perforation or hemorrhage.
PREVENTION:
There are 3 vaccines available for the prevention of typhoid fever:
Vivotif - a live attenuated oral vaccine administered to those over 6 years of age in three doses, one every other day.
Typhoid
vaccine VSP - a whole cell killed vaccine administered parenterally to
those over 6 months of age in 2 doses, 4 weeks
apart.
Typhim
Vi - a polysaccharide vaccine made up of the Vi antigen from the ty-2 strain
administered parenterally to patients 2 years
of age
or over in a single dose.
AMEBIASIS
OVERVIEW:
Infection is acquired by
ingestion of cysts of E. histolytica in contaminated food or water.
Excystation occurs in the small intestine, and trophozoites become established
in the lumen of the proximal colon in which they often live without causing
symptoms. Symptoms only result from invasion of the colon by trophozoites.
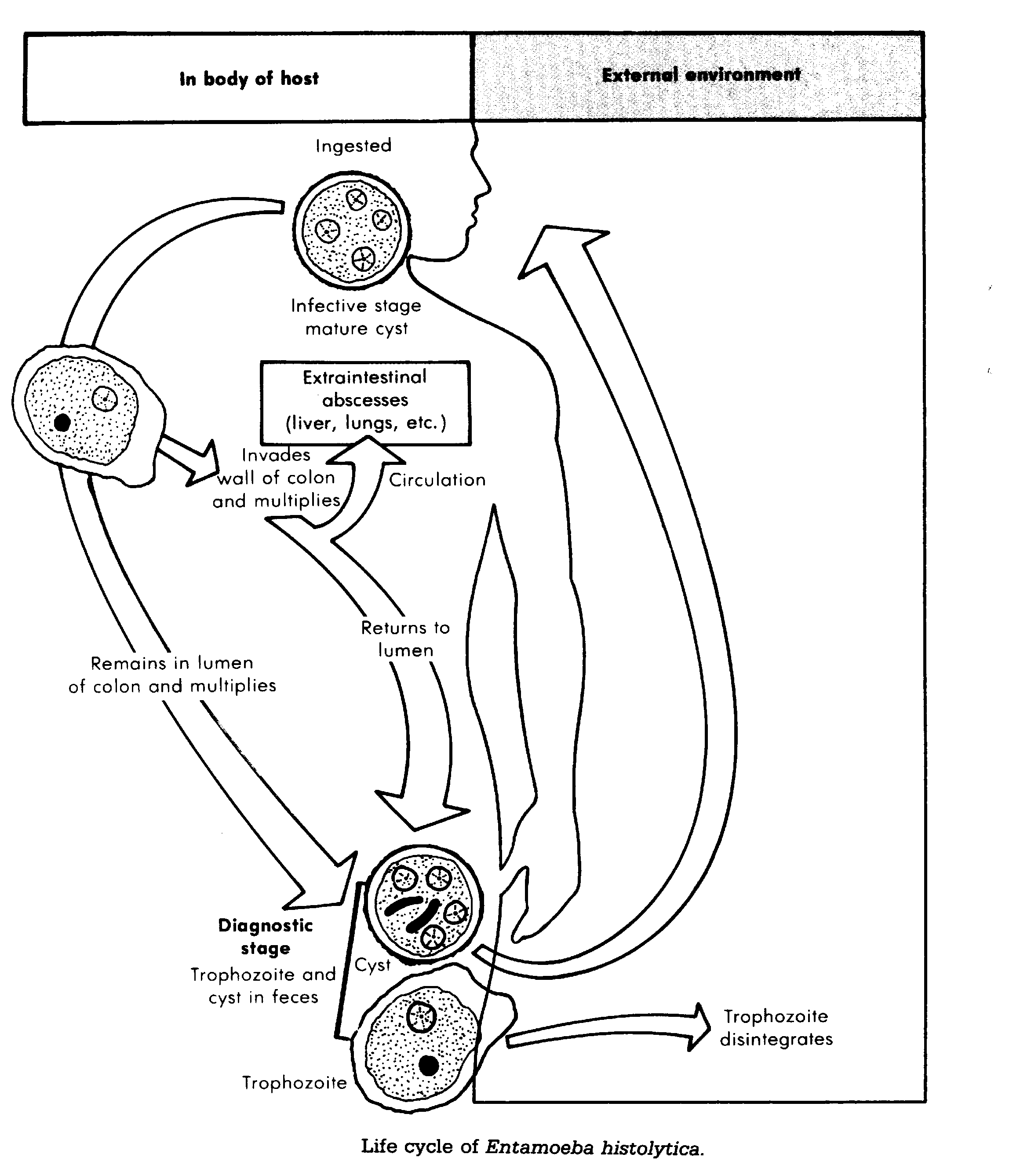
When mucosal resistance becomes lowered, trophozoites can invade colonic epithelium. Ameboid movement and amebic enzymes such as proteases, hyaluronidase and mucopoly-saccharidases facilitate penetration from the surface or within crypts of the mucosa. Eventually the venules and lymphatics are penetrated and the trophozoites gain access to the liver via the portal vein. The liver becomes the chief site of extraintestinal amebic disease.
Involvement of nonhepatic extraintestinal organs is much less frequent. Trophozoites may disseminate to other organs, especially from a liver abscess, by direct rupture into lung, pleural cavity or pericardium, or through the bloodstream to lung or brain. Cutaneous lesions may result from direct invasion of macerated epithelium in the perianal area when trophozoite-containing liquid feces contaminate the skin.
ETIOLOGICAL AGENT:
Entamoeba histolytica is the cause of amebiasis or amebic dysentery. It occurs in three stages: the active amoeba, the inactive cyst and the intermediate precyst. The ameboid trophozoite is the only form present in tissue. The cytoplasm is granular and may contain red blood cells (presence of intracytoplasmic red blood cells is pathognomonic).
PATHOLOGY:
The major lesions are colonic ulcers, colonic granulomas, diffuse hepatitis, hepatic abscess, brain abscess and rectal ulcerations. The primary lesions of the intestinal tract are ulcerations in the cecum, appendix and adjacent ascending colon. The unique feature of these ulcers is that they are flask-shaped. These lesions enlarge, gradually losing their characteristic form, and develop elevated margins with a white exudate on the base of the ulcer.
The organisms in the hepatic microcirculation produce necrosis of the endothelium and penetrate into the periportal sinusoids, where they may digest pathways into the hepatic lobules. Initially there is no inflammatory reaction but as necrosis progresses polymorphonuclear leukocytes gradually surround the lesion without formation of a definite wall. The lesions may remain focal or progress to form large solitary abscesses. Lung and brain abscesses are similar to those in the liver. Skin ulcerations show little inflammatory reaction.
SYMPTOMS:
After an incubation period of 1-5 days, the disease begins with a prodromal episode of diarrhea, abdominal cramps, nausea, vomiting and tenesmus; there may be vague abdominal discomfort, general malaise, loss of appetite, lose of weight and mental apathy. The feces may be watery or formed, but with dysentery they are generally watery, containing mucous and blood.
The association of an enlarged tender liver with fever and colitis is classic amebic hepatitis. Brain abscesses give rise to the symptoms of meningitis.
DIAGNOSIS:
The diagnosis of amebiasis is made by identifying E. histolytica in the feces or in tissues obtained from lesions or by the hela cell rounding assay for amebic cytotoxin..
TREATMENT:
The choice of amebicides is based on the location and severity of infection:
Asymptomatic
intestinal infection - diloxanide furoate (Furamide) or iodoquinol (Yodoxin)
or paromomycin (Humatin)
Necrotizing
amebic colitis or ameboma - diloxanide furoate plus metronidazole (Flagyl)
or dehydroemetine plus emetine
Extraintestinal
amebiasis - metronidazole plus iodoquinol.
Pleural
involvement may require drainage via a chest tube or thoracotomy.
CAMPYLOBACTERIOSIS
OVERVIEW:
C. jejuni is ingested with fecally-contaminated water, raw milk and food (especially poultry). It colonizes the jejunum where it elaborates:
Endotoxin
Enterotoxin
- similar to the LT toxin of Escherichia coli (causes diarrhea)
Verotoxin
- similar to the Shigella toxin (causes hemorrhagic colitis)
Three to five days after ingestion, overt disease occurs only if the organism penetrates the mucous coating the epithelial cells and invades the cell.
ETIOLOGICAL AGENTS:
Several species of Campylobacter cause gastrointestinal infections. C. jejuni is the most common cause of gastroenteritis worldwide. These are gram-negative, comma-shaped rods that commonly occur in pairs and are microaerophilic and motile.
PATHOLOGY:
The syndrome produced by C. jejuni is similar to that produced by Shigella and enteroinvasive Escherichia coli (EIEC).
SYMPTOMS:
Watery diarrhea is the most common manifestation. Clinical findings are not diagnostic. Along with the diarrhea there is sometimes pain, malaise and fever.
DIAGNOSIS:
Diagnosis is based on the finding of gull-shaped bacteria in watery, bloody, leukocyte-filled feces.
TREATMENT:
Ciprofloxacin may be useful
but this is generally a self-limited disease. Replacement of losses of
water and electrolytes, as in any diarrheal disease, is important.
ESCHERICHIA COLI INVASIVE GASTROENTERITIS
OVERVIEW:
The normal Escherichia coli carried in the human intestine has minimal or no invasive ability. A variant of E. coli that has been the recipient of gene transfer from Shigella sp. has gained the invasive abilities of Shigela sp. This E. coli variant is serotype 0157:H7. When ingested in undercooked meat it may cause hemolytic uremic syndrome, a potentially fatal disease.
ETIOLOGY:
E. coli 0157:H7 is a facultative, Gram-, motile, rod-shaped bacterium that cannot ferment sorbitol.
PATHOLOGY:
The majority of the pathology occurs in the ascending and transverse colon epithelium in the form of ulceration, edema, neutrophil infiltration, necrosis and exudation AND in the kidney in the form swollen glomerular epithelial cells, fibrin deposition and infiltrates of inflammatory cells.
SYMPTOMOLOGY:
The symptomology based on the intestianl pathology includes bloody diarrhea, abdominal cramps, dehydration and a mild fever. The symptomology attributed to the kidney pathology includes edema, dark urine and hypertension.
DIAGNOSIS:
The differential diagnosis should include:
Hemolytic
uremic syndrome
Shigellosis
Campylobacteriosis
Isolation and identification of the etiologic agent is necessary for definitive diagnosis.
TREATMENT:
The decision to treat or
not treat is controversial. Treatment results in the liberation of additional
toxin which can speed up kidney damage.
PEPTIC ULCER DISEASE
OVERVIEW :
The sources of Helicobacter pylori remain obscure. However, it is known that ingestion of 105 or larger number of organisms will cause peptic ulcer disease. By five to ten days after the ingestion, the organisms have penetrated the stomach epithelium with production of gastritis.
ETIOLOGICAL AGENT:
Helicobacter pylori. This is a curved, rod-shaped bacillus that is Gram-negative but stains best in tissue biopsies using a modified Giemsa stain. It is motile, microaerophilic and a slow grower (it takes at least 7 days to form a colony on agar medium at 37°C). It contains an adhesin which is also a hemagglutinin. Its most unique characteristic is copious production of urease.
PATHOLOGY:
The epithelial cells of the pylorus of the stomach are the primary target of chronic infection that may result in chronic active gastritis and peptic ulceration. This correlates with adenocarcinoma of the stomach. Ammonia from urea and nicotine potentiate the action of the action of a cytotoxin produced by H. pylori. The end result is vacuolization of the epithelial cell and induction of a chronic inflammatory process and eventual ulcer formation. The inflammatory response is primarily one of mononuclear cells.
CLINICAL SYMPTOMS:
Patients with peptic ulcer disease have an associated gastritis. Gastritis results in dyspepsia, cramps, halitosis, ructus, nausea, vomiting and flatulence.
DIAGNOSIS:
Antibodies specific for H. pylori is suggestive of helicobacteriosis. Histological identification of H. pylori in gastric biopsy specimens buttressed by isolation in culture is confirmative.
TREATMENT:
Combination therapy, consisting
of antimicrobics and bismuth-containing drugs, is effective in eradicating
H.
pylori from the stomach. However, the optimum therapy has not yet been
defined. Suggested treatment presently is omeprazole + amoxicillin + bismuth
or omephrazole + amoxicillin + clarithomycin.
CROHN'S DISEASE
NAMES OF DISEASE:
Crohn's Disease
Regional enteritis
OVERVIEW:
While the etiology of Crohn's disease is still somewhat controversial, there is solid evidence that the disease is caused by ingestion of undercooked meat containing Mycobacterium paratuberculosis. This leads to granulomatosis and ulceration of the terminal ileum and colon which gives rise to fever, adbdominal pain, diarrhea and fatigue.
ETIOLOGICAL AGENT:
Mycobacterium paratuberculosis, a Gram+, acid-fast+, plump rod.
EPIDEMIOLOGY:
This organism causes Johne's Disease in cattle and sheep. The organism infects the intestinal mucosa of cattle and sheep, causing a chronic diarrhea. It is transmitted to humans via ingestion of undercooked cattle/sheep intestines.
PATHOLOGY:
After ingestion of the organism it invades the human intestinal mucosa in the terminal ileum and the colon. A chronic inflammation extends through all layers of the intestinal wall and involves the mesentery as well as regional lymph nodes. The bowel appears greatly thickened and leathery with the lumen narrowed. This may cause varying degrees of intestinal obstruction. The mesentery is thickened, fatty, and often extends over the serosal surface of the bowel in characteristic finger-like projections. As the infections progresses the tissue, becomes granulomatosis. There is characteristic fistula and abscess formation. The disease is often discontinuous with patches of infected tissue separated by areas of normal tissue. The ulcers are linear and the granulomas are non-caseating.
SYMPTOMOLOGY:
The symptoms include fever, abdominal pain, diarrhea, generalized fatigability and the severe anorectal complication of fistulas, fissures and perirectal abscesses.
DIAGNOSIS:
Diagnosis is generally achieved by fiber optic colonoscopy for tiny longitudinal fissures or ulcers and a cobblestone appearance. Isolation of Mycobacterium paratuberculosis requires specialized cultural conditions, is difficult and unreliable and is not routinely done. M. paratuberculosis DNA can be detected in colon biopsies via the polymerase chain reaction (PCR).
TREATMENT:
Antimicrobial treatment has
not been universally effective. Possibilities for treatment include sulfasalazine
and metronidazole. Commonly used anti-tuberculosis drugs have not proven
effective.