MM 245-249,236-237,622-624;
ID 733-759,769-771, 736-738
INFLAMMATORY GASTROENTERITIS
CHOLERA
ETIOLOGICAL AGENT:
Vibrio cholerae. This is a slightly curved gram-negative rod which has two major groups based on the O-antigen. These are identified by slide agglutination tests with specific antiserum. Classic epidemic cholera is caused by the 01 serotype; all other strains are designated the non-01 strains and they have the antigen designations 02-0139. These non-01 strains produce sporadic and milder forms of diarrhea.
OVERVIEW:
Cholera is endemic in India, West Bengal, Bangladesh and Louisiana in the U.S. The organism is ingested with water or food (especially shellfish and crabs) and causes an acute illness due to an enterotoxin elaborated by V. cholerae that have colonized the small bowel. In its most severe form, there is rapid loss of liquid and electrolytes from the gastrointestinal tract, resulting in hypovolemic shock, metabolic acidosis and, if untreated, death.
PATHOLOGY:
V. cholerae is very acid sensitive and the majority of ingested organisms are killed by stomach acidity; it takes ingestion of 108-1010 cells to cause disease. Those few organisms that survive stomach and upper intestinal acidity attach to the microvilli of the glycocalyx of epithelial cells of the jejunum and ileum. There they multiply and liberate cholera enterotoxin, mucinase and endotoxin. They do not invade the mucosa. All signs, symptoms and metabolic derangements in cholera result from the rapid loss of liquid from the gut. The feces are nearly isotonic with plasma. As compared with plasma, the concentrations of sodium and chloride are slightly less, bicarbonate is twice as high, and potassium is 3-5 times higher. The increased electrolyte secretion is caused, in the absence of morphologic damage to the gut mucosa, by a protein enterotoxin coded for by a chromosomal gene. The enterotoxin has a molecular mass of 84,000 daltons and consists of a binding (B) moiety and an activating (A) moiety.
Five equal subunits with
a molecular weight of 11500 each make up the B moiety.
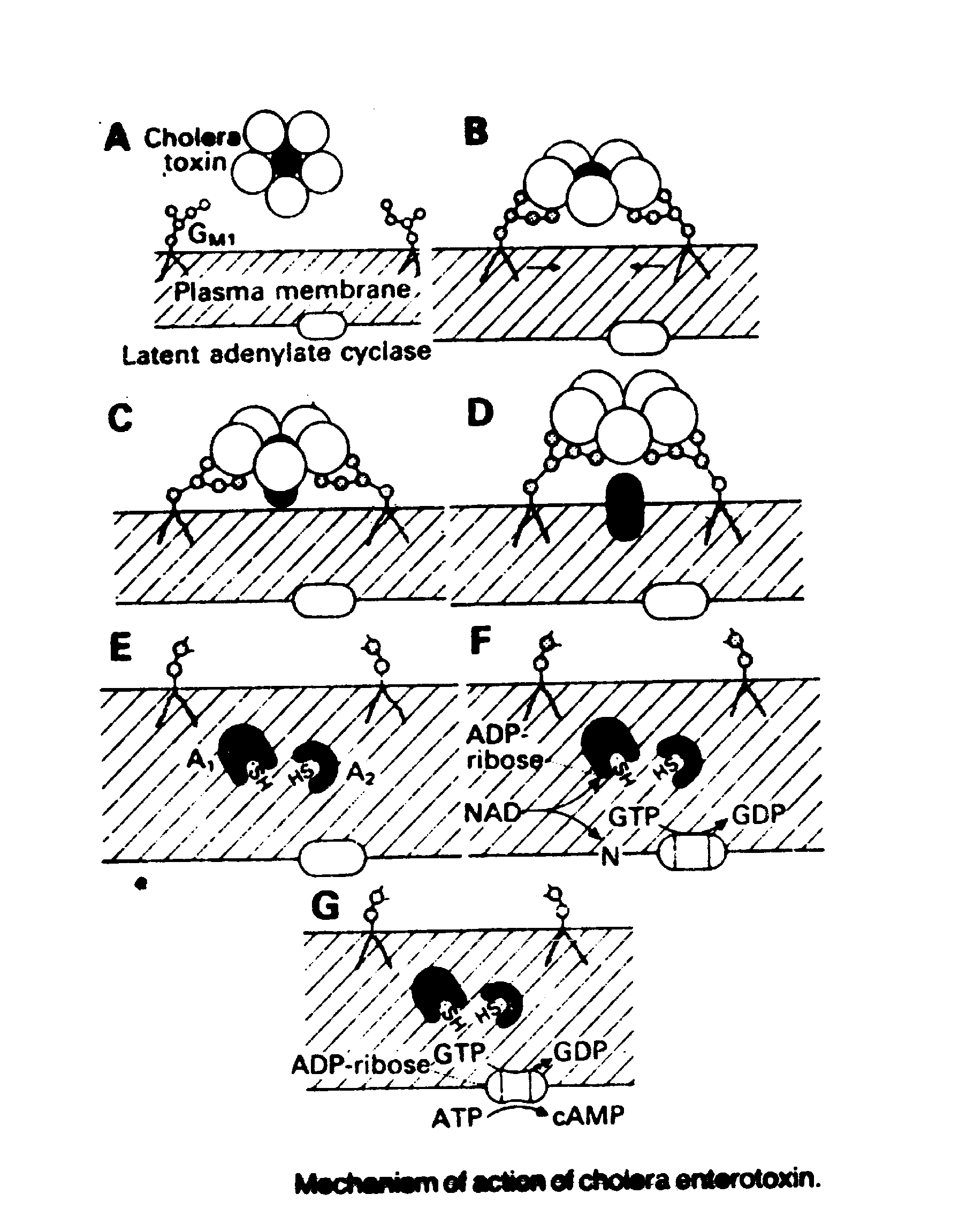
On exposure to small bowel epithelial cells, each B subunit rapidly binds to GM1 monosialoganglioside in the gut cell wall. Following binding, the A moiety (composed of two unequal subunits) migrates through the epithelial cell membrane. The A1 subunit contains ADP-ribosyltranferase activity and catalyzes the transfer of ADP-ribose from NAD to a guanosine triphosphate (GTP) - binding protein that regulates adenylate cyclase activity. The ADP-ribosylation of GTP binding protein inhibits the GTP turnoff reaction and causes a sustained increase in adenylate cyclase activity. The resultant increased intracellular cyclic AMP acts at 2 sites to cause net secretion of isotonic liquid within the small bowel lumen. The increased cyclic AMP inhibits neutral sodium chloride absorption across the glycocalyx via the cotransport mechanism; it also stimulates active chloride secretion into the gut lumen. There is no significant pathology.
CLINICAL SYMPTOMS:
The onset is characterized by abrupt, watery diarrhea. Several liters of liquid may be lost within a few hours, rapidly leading to profound shock. Vomiting may ensue after diarrhea. The patient is cyanotic and has sunken eyes and cheeks, a scaphoid abdomen, poor skin turgor and thready or absent peripheral pulses. The voice is high pitched or inaudible; the vital signs include tachycardia, tachypnea and low or unobtainable blood pressure. The heart sounds are distant and often inaudible, and bowel sounds are hypoactive.
DIAGNOSIS:
In endemic or epidemic areas, the working diagnosis of cholera is made based on the clinical presentation, especially the presence of "rice water" stools. Confirmative diagnosis is made by plating a stool sample on TCBS (thiosulfate-citrate-bile salt-sucrose) agar, which is selective for Vibrio, and the adrenal cell assay.
TREATMENT:
Successful therapy requires only prompt replacement of fluids and electrolytes. Ringer's solution is most commonly used. It is given rapidly by IV injection - 50 to 100 ml per minute - until a strong radial pulse is restored. Tetracycline reduces the severity and length of disease. Chloramphenicol and furazolidone are slightly less effective.
PREVENTION:
A parenterally administered
vaccine containing 109 killed vibrios per ml is available. Protection
lasts for 3-6 months. It is effective only against 01 serotypes.
ESCHERICHIA COLI INFECTION
ETIOLOGICAL AGENT:
Enterotoxigenic E. coli (ETEC) or enteropathogenic E. coli
(EPEC)
PATHOLOGY:
EPEC produces no demonstrable toxin but most strains are enteroadherant (EAEC) and cause alterations in the glycocalyx of the small bowel epithelial cells. EPEC express rope-like bundles of filaments, termed bundle-forming pili, that create a network of fibers that bind together the individual organisms. The EPEC pili are homologous with toxin-coregulated pili of Vibrio cholerae. The gene for these fibers is plasmid-borne. ETEC has multiple pathogenic mechanisms which include at least 2 distinct toxins; a heat-labile toxin (LT) and a heat stable toxin (ST). The ability to produce enterotoxin is mediated by a single plasmid. The E. coli LT is similar, but not identical to, the cholera toxin; it binds to the same site, has the same effect and serologically cross-reacts with cholera toxin. Like cholera toxin it will bind to cells outside the intestine.
The E. coli ST is quite different from the LT because it exhibits a rapid onset of action, does not bind to gangliosides of the mucosal cell membrane, and is of low molecular weight, that is, less than 2000 daltons. It is not antigenic. The ST acts by stimulation of guanylate cyclase with resultant cyclic GMP accumulation in mucosal cells. It only affects cells of the small intestine. The kinetics of ST action are strikingly different from LT action, because it causes an almost immediate increase in the secretion of gut fluid. The increase in intracellular guanylate cyclase causes chloride secretion by gut mucosal epithelial cells in a manner similar to that seen with the cholera enterotoxin, but the ST does NOT alter neutral sodium chloride absorption by the brush border cotransport route. ST binding is specific for small bowel mucosal cells and thus, unlike cholera enterotoxin and E. coli LT, it affects only intestinal cells. Besides toxin production, the major other violence factors that EPEC and ETEC produce are colonization factors which allow them to adhere to mucosal cells. These are various types of fimbriae or pili. These strains produce little or no gross pathology.
SYMPTOMS:
Severe diarrheal disease caused by ETEC is generally characterized by the abrupt onset of watery diarrhea. In severe cases, the clinical picture is identical to that of cholera except that cramping abdominal pain is more commonly present with E. coli diarrheas and the duration is much less, seldom lasting more than 24 hours after initiation of fluid replacement therapy. The non-enterotoxin producing, noninvasive E. coli (EPEC) have thus far been incriminated only in relatively mild diarrheal disease in infants and small children.
DIAGNOSIS:
Diagnosis is made by isolating E. coli on MacConkey's agar and then:
1. Inoculating
them into a tissue culture of mouse adrenal cells or Chinese hamster ovary
cells which respond morphologically to
stimulation of their adenylate cyclase systems by the LT or
2. Performing an ELISA test on toxin bound to antibody or
3. Using
a DNA probe to detect the LT gene.
TREATMENT:
Intravenous or peroral replacement
of the fluid and electrolytes lost in feces. Peroral therapy is almost
always adequate. Tetracycline and trimethoprim - sulfamethoxazole are effective
in shortening the duration of symptoms but are not essential. Bismuth subsalicylate
may provide symptomatic relief (less severe abdominal cramps and less frequent
stools).
GIARDIASIS
NAME OF DISEASE: Giardiasis
OVERVIEW:
Ingestion of water containing a Giardia lamblia cyst results in the cyst developing into a trophozoite in the duodenum. The trophozoites in the duodenum and jejunum destroy the glycocalyx causing a malabsorption and diarrhea.
ETIOLOGICAL AGENT:
Giardia lamblia, a flagellate with both a trophic and cystic stage. The teardrop-shaped trophozoites have a smooth dorsal surface with a concave ventral surface and a prominent anterior adhesive disk. There are 4 pairs of flagella directed posteriorly. The cysts are ellipsoidal and highly refractile. This is the most common intestinal protozoan parasite of humans.
PATHOLOGY:
Ingested organisms colonize the duodenum and jejunum where they injure the epithelium of the microvillus, which is manifested as a functional derangement in the cell membrane and a loss of the glycocalyx coat. Loss of the digestive enzymes and transport mechanisms for mono-saccharides and amino acids leads to malabsorption and diarrhea. Pathologic changes are mild in most cases, but shortening and thickening of the villi associated with acute focal inflammatory changes in the mucosal epithelium may be seen initially and are followed by chronic inflammatory infiltrates in the lamina propria.
SYMPTOMOLOGY:
After a one week incubation period the patient may be asymptomatic or exhibit abdominal cramps, diarrhea, anorexia, nausea, malaise and abdominal bloating. A sulfur-tasting belching is common. Symptoms spontaneously resolve in 4-6 weeks.
DIAGNOSIS:
Presumptive diagnosis is made on the basis of a history of drinking non-chlorinated water and the expression of classical clinical symptoms. Confirmative diagnosis requires the finding of Giardia lamblia trophozoites or cysts in the feces or in an intestianl biopsy.
TREATMENT:
Quinacrine hydrochloride
(Atabrine; 100 mg, PO, every 8 hours for 10 days) or metronidazole (Flagyl;250
mg, PO, every 8 hours for 10 days).
ANTIMICROBIC-ASSOCIATED PSEUDOMEMBRANOUS COLITIS
OVERVIEW:
Clostridium difficile is a bacterium that is resistant to most antibiotics. It is present in the intestine of about 5% of humans. Long term antibiotic therapy, especially with clindamycin, reduces the number of viable bacteria in the intestine but allows C. difficile to become the predominate organism in the GI tract. The organism produces small amounts of toxin A and toxin B which only achieve cytotoxic levels when it is the predominant organism. Intestinal epilhelial cells are killed, forming a pseudomembrane under these conditions.
ETIOLOGY:
Clostridium difficile is an anaerobic, Gram-positive, spore-forming rod that produces toxin A and toxin B. Both toxins are cytophilic but only toxin A is active against intestinal epithelial cells.
PATHOLOGY:
The major pathology occurs in the lower reaches of the ileum and in the colon. The pathology begins with small white/yellow plaques that, with time, enlarge and develop a hemorrhagic border. These areas of epithelium become necrotic and slough off forming a pseudomembrane with inflammatory exudate.
SYMPTOMOLOGY:
The common symptoms include fever, diarrhea, abdominal distension and abdominal pain.
DIAGNOSIS:
Diagnosis is generally made on the basis of a history of antibiotic therapy within the past month, especially a history of clindamycin, ampicillin or cephalosporin therapy and on the presentation of the symptoms listed above. Colonoscopy revealing the classical pathology can add weight to the diagnosis. The feces can be tested for the presence of the etiological agent and/or the presence of toxin A.
TREATMENT:
Withdrawal of the antibiotic
and replacement of the intestinal flora generally suffices. If antibiotic
treatment is needed, vancomycin or metronidazole will kill C. difficile.
VIRAL GASTROENTERITIS
OVERVIEW:
The two major causes of human viral gastroenteritis (the rotavirus and the Norwalk virus) are carried in the intestine of most domestic and many wild animals. Fecal-oral spread from animal-to-person or person-to-person causes a diarrhea. Rotavirus disease (winter diarrhea) is most common among infants and young children while Norwalk virus disease (summer diarrhea) affects older children and adults.
ETIOLOGY:
The rotavirus is in the Reoviridae family, i.e., it is a naked double-stranded RNA virus with a double icosahedral capsid.
The Norwalk virus is in the Caliciviridae family, i.e., it is a naked single stranded RNA-containing virus with a single icosahedral capsid.
PATHOLOGY:
Rotaviruses and Norwalk viruses invade and destory mature epithelial cells in the middle and upper villus, causing a decreased absorption of sodium and water from the bowel lumen.
SYMPTOMOLOGY:
After a 2-day incubation period there is diarrhea, nausea and vomiting.
TREATMENT:
Viral gastroenteritis is a self-limiting disease but it is often necessary to adminster fluids and electrolytes.