Syllabus | Next Lecture
MM 719-722
![]()

2. To introduce the concept of the "prion" as an etiological agent of human disease.
3. To introduce the concept of the "viroid" as a disease entity.
4. To familiarize you with the current concepts of the structure, replication and pathogenicity of the prion and viroid.
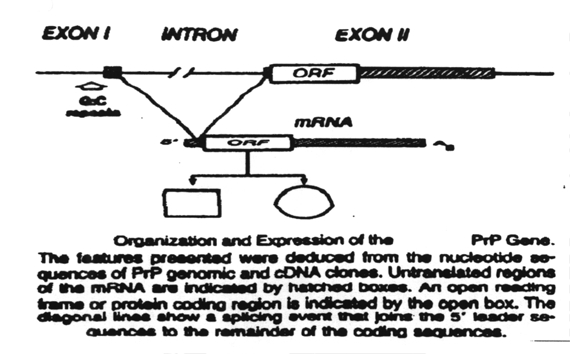
The PrP 33-35 undergoes several post-translational events to become the prion protein (PrP 27-30):
2. Formation of a disulfide bond between two cysteine residues.
3. Removal of the N-terminal signal peptide.
4. Removal of the C-terminal hydrophobic segment.
5. Addition of a phosphatidylinositol glycolipid at the C-terminal.
6. Removal of the N-terminal first 57 amino acids.

2. The b-sheet-forming peptides aggregate to form amyloid fibrils.
3. The amyloid fibrils kill thalamus neurons through apoptosis, a programmed series of events that leads to cell death.
2. Depletion of dendritic spines in neurons.
3. Formation of numerous vacuoles in the cerebellar cortex (spongiform encephalopathy).
4.
Amyloidosis - deposition of amyloid in the cerebellar cortex, thalamus,
brain stem and in the lumen of blood vessels
within the brain. These amyloid plaques consist of discrete eosinophilic
glassy-appearing masses, often having
radiating amyloid fibrils at their periphery. The plaques are primarily
subependymal, subpial and perivascular.
These pathologies give rise to the clinical symptomology seen in these patients. These are:
1. A long incubation period (several years) which has given rise to the term "slow infection."
2. Loss of muscle coordination which leads to a difficulty in walking, indicating a functional disorder of the cerebellum.
3. Dementia characterized initially by loss of memory, diminished intellect and poor judgement.
4. Progressive insomnia characterized by a marked reduction or loss of the slow-wave and rapid-eye-movement phases.
Spread of the disease is via horizontal transmission, i.e., transmission from one person to another, either directly or by fomites or by ingestion of contaminated meat.
In the past, diagnosis of prion disease was made through examination of brain biopsies taken from patients in advanced stages of the disease or, more commonly, after they had died. In January of 1999 it was found that the prion protein accumulated in the tonsils and could be detected by an immunofluorescence test on tonsilar biopsies. A second test was simultaneously developed which was based on a Western blot. Later that year a third test was developed that had the high sensitivity necessary to detect the prion protein in blood. This test is based on capillary electrophoresis with laser-induced fluorescence. It detects as little as 10-18 mole.
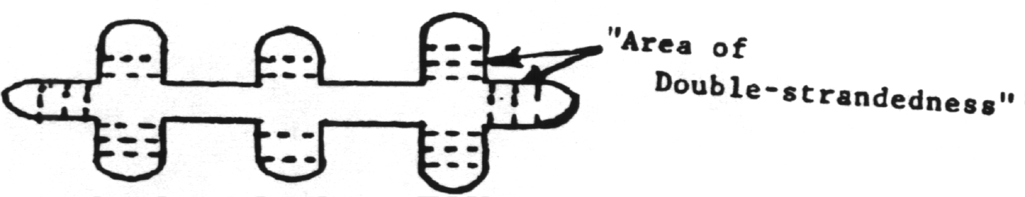
Because of their simplified structures both prions and viroids are sometimes called subviral particles. Viroids mainly cause plant diseases but have recently been reported to cause a human disease.
Catalytic RNAs are those that have the intrinsic ability to break and form covalent bonds; Viroids are catalytic RNA's (ribozymes) that cleave RNA to produce fragments containing a 5'-hydroxyl and a 2', 3'-cyclic phosphate.
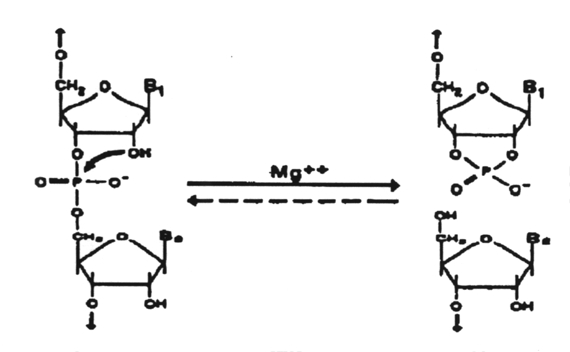
This is a nonhydrolytic reaction in which the same number of phosphodiester bonds are maintained and the transesterification reaction is theoretically reversible. This reaction is considered to play an essential role in the replication of these RNAs in vivo. Such reactions are all intramolecular and hence quasi-catalytic with single turnover. These RNAs can be manipulated, however, to provide true catalytic cleavage in trans-reactions.
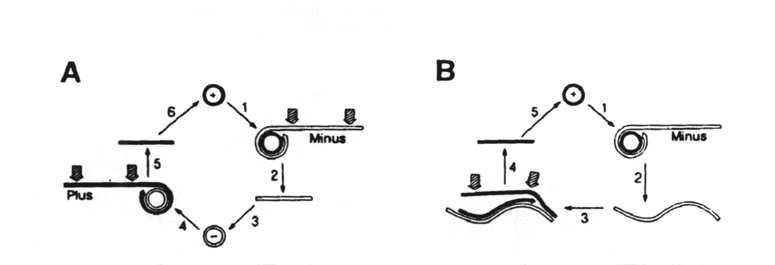
In the first variation (A), the circular plus strand is copied by viroid RNA-dependent RNA polymerase to form a concatameric minus strand (step 2). Site-specific cleavage (arrows) of this strand produces a monomer that is circularized by a host RNA ligase (step 3) and then copied by the RNA polymerase to produce a concatameric plus strand. Cleavage of this strand (step 5) produces monomers which, on circularization, produces the progeny circular, plus RNA, the dominant form in vivo.
In the other variation (B), the concatameric minus strand of step 1 is not cleaved but is copied directly to give a concatameric plus strand (step 3), which is cleared specifically to monomers for ligation to the circular progeny. Those RNAs that self-cleave only in the plus strand in vitro are considered to follow this route.
The hepatitis D viroid genome is a minus strand that gives rise to two RNA species. One of these is a mRNA for the delta antigen and the other is a complete complimentary copy (plus strand or anti-genome). The anti-genome acts as a template to make more minus strands. The minus strand self-cleaves and self-ligates. HDV replication takes place in the nucleus but delta antigen is made in the cytoplasm. The delta antigen is the only protein made by the HDV mRNA. It has a +12 charge at physiologic pH, accumulates in the nucleus and binds to minus strand RNA as a dimer. The delta antigen is necessary for viroid assembly but its exact mode of action is unknown.
2. Prions are infectious agents composed solely of glycoprotein. They are products of a human gene which accumulate in tissue as amyloid.
3.
Amyloid deposition in tissue is a pathological manifestation of many diseases,
of both prion and non-prion etiology. These
diseases include Alzheimer's disease, *Creutzfeldt-Jakob disease, Down's
syndrome (mongolism), *fatal familial insomnia,
*Gerstmann-Straussler syndrome, *kuru and leprosy. (* indicates prion diseases).
4. The accumulation of amyloid induces these pathologies in the host: astrocyte gliosis, depletion of neuronal dendritic spines, spongiform encephalopathy.
5.
In prion disease there is a long incubation period before one sees loss
of muscle coordination, dementia and/or progressive
insomnia.
6. Prions induce no immune reactions within the human.
7. There are 3 tests available for the diagnosis of prion disease: immunofluorecence test on tonsil biopsies, Western blot analysis of blood tissue, capillary electrophoresis of blood.
8. Viroids are infectious agents composed solely of circular single-stranded RNA which folds over on itself to form some double stranded regions. These are catalytic RNAs (ribozymes).
9. The viroid RNA codes for a single protein, the delta antigen.
10. The only human disease known to be caused by a viroid is hepatitis D; in this case the viroid is enclosed in a hepatitis B virus capsule.
11. The hepatitis D viroid manifests its disease potential by sequestering and/or destroying human liver 7S RNA.